With roots dating back as far as 2006, the Society of Interventional Oncology (SIO) is the only nonprofit, member-based organization dedicated completely to interventional oncology (IO). SIO seeks to establish IO as the fourth pillar of cancer therapy, alongside medical, surgical and radiation oncology, by developing evidence supporting IO therapies, educating IO practitioners and improving patient access to IO therapies.

Given the increasing importance of minimally invasive IO procedures for selective delivery of treatments to tumors (such as chemotherapy, radioactive particles or immunotherapy for liver cancer), thermal ablation to destroy cancer cells, and palliative care, TriSalus enthusiastically supports SIO and IO professionals as an SIO2023 Premiere Industry Partner.
Five ways SIO advances IO and supports IO professionals
1. IO University: SIO maintains a robust on-demand educational resource as a way for IO professionals to build skills, access best practices and earn CME credits online throughout their career. The curriculum spans topics across diagnosis, disease state and treatment modalities — from the fundamentals to expert insights.
2. IO Awareness Week: Organized by SIO, this annual campaign spreads the word about the growing IO field and the benefits of IO therapies for patients with cancer through conversations on social media, sharing real patient stories, complimentary webinars and podcasts on trending IO topics, and a range of resources for IO professionals and industry partners to participate in.
3. National Comprehensive Cancer Network (NCCN) Guidelines: SIO submits NCCN guideline requests that support the evolving IO practice and has successfully elevated guidelines for multiple solid tumors (including hepatocellular carcinoma, colorectal cancer and renal cell carcinoma), deep vein thrombosis and palliative care.1,2
4. IO Essentials Program & Scholarships: This program is designed by SIO to help early-career interventional oncologists build a successful practice, network with IO peers and pioneers and become the future of the field. This year, the scholarship program — supported by industry partners, including TriSalus — will help more than 50 fellows and residents-in-training to attend SIO2023, enjoy SIO membership and access the IO Essentials educational programming.3
5. Clinical Research, Trials and Grants: Robust and ongoing clinical research is indispensable to the advancement of IO procedures and outcomes. SIO has awarded over $1.5 million to support preclinical research to date, has sponsored major clinical trials and has been instrumental in developing trial concepts.4 In partnership with the Society of Interventional Radiology (SIR), SIO founded the Global Interventional Oncology Clinical Outcomes Registry (IO-CORE) to create a large-scale repository of prospective clinical data, providing a broad view of disease management and outcomes across IO therapeutic strategies.
SIO’s premier annual event
The SIO Annual Scientific Meeting is SIO’s premier annual event. It is a forum for IO professionals from around the globe to network, share patient care practices, advance knowledge and skills and learn about the latest innovations and clinical research.
The SIO2023 Annual Scientific Meeting (January 19-23, Washington DC) will offer five days of valuable programming, including master classes, broad-based learning topics, interactive organ-based discussions, case-based reviews, and featured presentations on basic, translational and clinical research.
How does TriSalus support SIO and IO professionals?
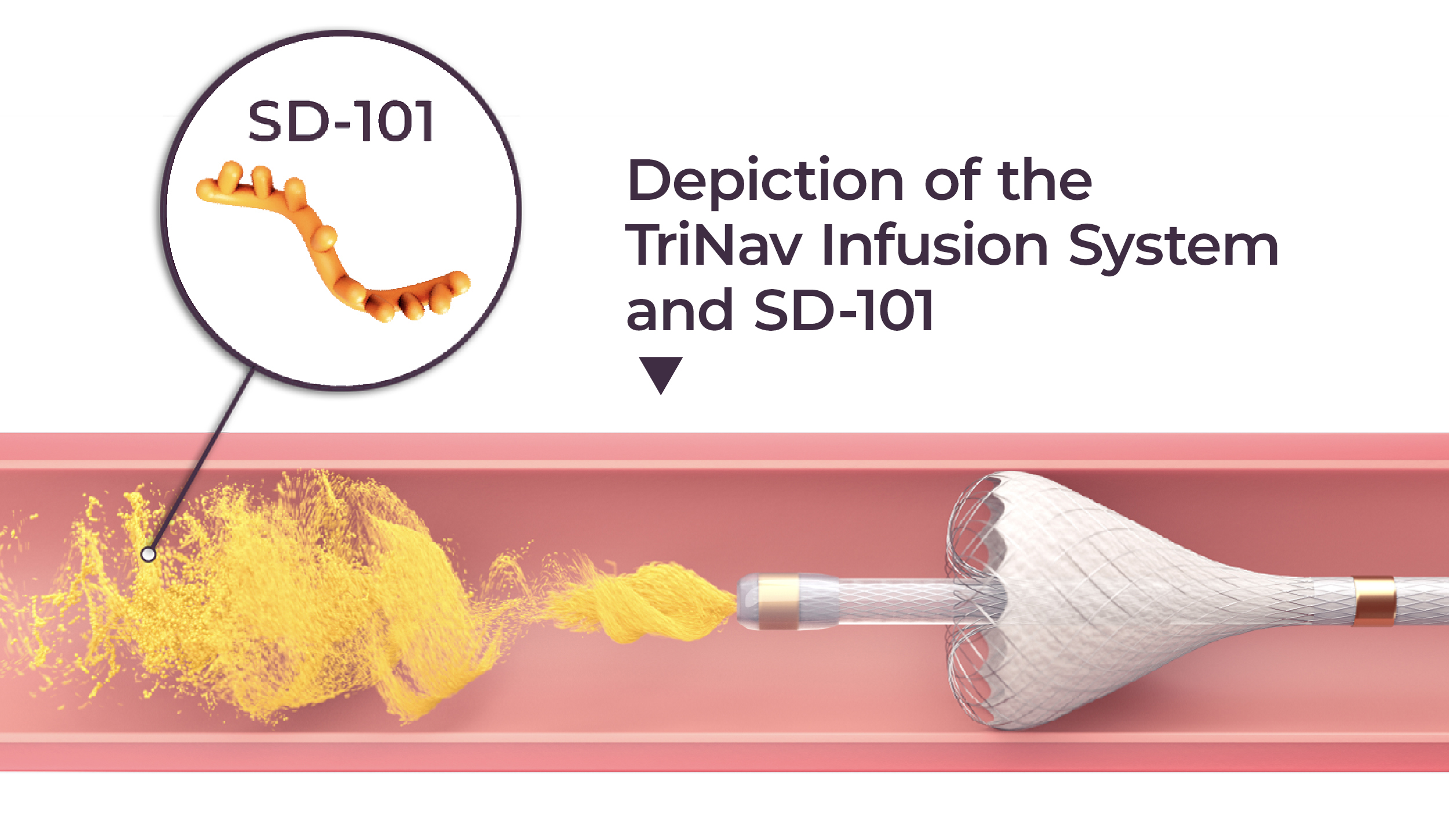
SD-101 is an investigational drug and has not been approved by FDA or any health authority globally.
TriSalus is committed to advancing the treatment and transforming the lives of patients with difficult-to-treat liver and pancreas tumors. Our commitment does not just live in words — it is evident in our support of the professionals who are helping to expand the science around tumor treatment.
During SIO2023, on January 21, TriSalus is sponsoring an educational lunch symposium — Seeking to Enable Immunotherapy for Liver and Pancreatic Tumors: Updates from PERIO Trials — where the latest updates on the ongoing Pressure-Enabled Regional Immuno-Oncology (PERIO™) clinical trials will be presented. These studies are testing the TriSalus immunotherapy platform, which integrates SD-101 — an investigational immunotherapy — and the Pressure-Enabled Drug Delivery™ (PEDD™) approach using the TriNav Infusion System for delivery to liver tumors in adults with metastatic uveal melanoma, hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (ICC).
Clinical trials are especially important for patients who have rare conditions or unmet medical needs — such as HCC, ICC and metastatic uveal melanoma. If you are aware of patients who might benefit from taking part in these or other trials, find out more about eligibility criteria at ClinicalTrials.gov.
SD-101 is an investigational drug and has not been approved by FDA or any health authority globally.
Rx Only. For the safe and proper use of the TriNav device, refer to the Instructions for Use.
Indications for Use: The TriNav Infusion System is intended for use in angiographic procedures. It delivers radiopaque media and therapeutic agents to selected sites in the peripheral vascular system.5
Contraindications: TriNav is not intended for use in the vasculature of the central nervous system (including the neurovasculature) or central circulatory system (including the coronary vasculature).5
REFERENCES:
1. Why you should become an SIO Member. Accessed November 22, 2022. https://youtu.be/2WjyWfTjxSY
2. NCCN Guidelines. Society of Interventional Oncology. Accessed November 22, 2022. http://www.sio-central.org/page/nccn
3. IO Essentials. Society of Interventional Oncology. Accessed November 22, 2022. http://www.sio-central.org/page/io-essentials-818
4. Research and Grants. Society of Interventional Oncology. Accessed November 22, 2022. http://www.sio-central.org/p/cm/ld/fid=845
5. TriSalus TriNav Infusion System, Instructions for Use.